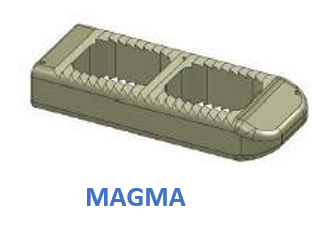
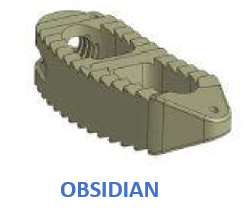
Blustone Synergy has created an advanced line of PEEK and Titanium Coated PEEK Interbody spacers for the Cervical and Thoracolumbar Spine. We have combined years of engineering and medical experience to provide FDA cleared devices that enhance the surgical experience. We have tapered and radius'ed our cages to allow for larger graft volumes and bigger fusion surface areas. We offer an added 10 degree hyper-lordosed lumbar lateral cage for better deformity correction when needed most. Our TLIF cage can be inserted with one instrument to fully rotate the cage 90 degrees into its optimal location, minimizing the need to place additional instrumentation adjacent to the spinal cord. The TLIF cage in its final position provides lordosis to best match patient anatomy in the lumbar spine. We manufacture a 5mm cervical cage that surpasses mechanical testing to withstand anatomical forces, for the most challenging ACDF surgery. Our vertebral body replacement device is lordosed in 3 different configurations, 20 to 50mm heights, and made from one solid PEEK bar to resist the high loads and stress. By adding our Lavaflow, advanced Plasma Sprayed Titanium, to our existing lineup of interbody devices, we have enhanced the properties of our existing PEEK Interbody Spacers. This thin coating of Titanium offers additional osseointegration benefits while maintaining the same great radiographic properties and modulus of elasticity of PEEK. Combining these two materials brings together two proven technologies to enhance patient outcomes for the future.
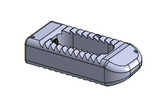
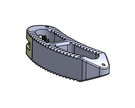
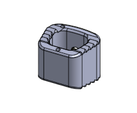
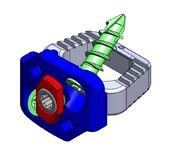
The release of Diamond SA Cervical System expedites the Anterior Cervical Fusion Procedure while minimizing tissue disruption with this low profile implant. The Integrated Cervical Interbody and Fixation System advanced technology meets surgeon and patient needs for a sleeker streamlined surgery.
These are just a few of the additional features our products offer among many to aid in the surgeon's hands and streamline the spinal surgical procedure. Contact your spine sales representative or our company for more information on product features, benefits, and availability. Blustone Synergy's products presented on this website have been cleared by the FDA.
The release of Diamond SA Cervical System expedites the Anterior Cervical Fusion Procedure while minimizing tissue disruption with this low profile implant. The Integrated Cervical Interbody and Fixation System advanced technology meets surgeon and patient needs for a sleeker streamlined surgery.
These are just a few of the additional features our products offer among many to aid in the surgeon's hands and streamline the spinal surgical procedure. Contact your spine sales representative or our company for more information on product features, benefits, and availability. Blustone Synergy's products presented on this website have been cleared by the FDA.